An innocuous bottle of bathroom cleaner sits on the Ubatuba vanity top. “Oh, I’ll just put that away,” you muse as you grab the bottle and proceed to the laundry room. But wait…
Underneath the bottle is a doughnut-shaped ring of white, standing out like a sad constellation against the black surface. When the liquid was poured from the bottle of cleaner, a few remnant drips seeped down the outside of the container and collected underneath the bottle. The liquid sat on the stone, and an unfortunate chemical reaction ensued.
What happened? What can be done? And how can this be prevented? Read on for the full scoop on how acids can damage even the toughest natural stones…
What’s in Toilet Bowl Cleaner?
When it comes to cleaning toilets, most of us prefer to let a strong chemical do the work rather than personally engaging in the act of deep scrubbing. Thus, we’ve come to tolerate the use of potentially damaging chemicals in our homes. The active ingredient in many toilet bowl cleaners is hydrochloric acid. Some cleaners use citric acid or phosphoric acid. In any case, the acid dissolves hard water, rust, and soap scum – and that means we don’t have to scrub very hard to get rid of stains.
You can look up the details of any cleaning product by going to the product’s official website and looking for the MSDS – Material Safety Data Sheet. It’s an excellent way to learn exactly what you’re dealing with. For example, here’s the safety data sheet for Lysol Power toilet bowl cleaner, with some key excerpts in the image below.
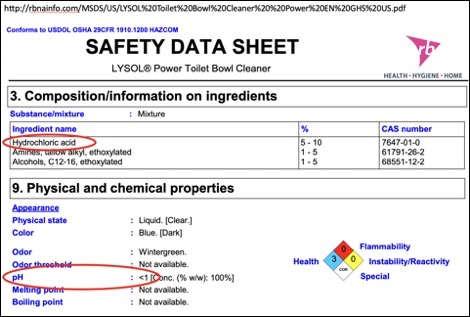
Remember the pH Scale?
Toilet bowl cleaner is a strong acid. How strong? Well, the pH of Lysol toilet bowl cleaner is less than one. You might wonder how it’s even possible to have a pH that low, but the pH scale doesn’t stop at one or even zero; pH can actually be a negative number.
In the highly likely event that you forgot this tidbit from high school chemistry, pH stands for the potential of hydrogen. A low pH is acid, seven is neutral, and above seven is alkaline. An acidic solution has extra hydrogen ions floating around in it. The more hydrogen ions, the stronger the acid. Each number on the pH scale represents a ten-fold difference in the acidity. So a cleaning product with a pH of 1 is ten times more acidic than one with a pH of 2. If you’d like to refresh your memory a bit more, here’s a short article about pH, which lists the pH of common liquids.
Strong Acids Affect Nearly All Rocks and Minerals
It’s well understood that acids etch limestone, marble, dolomite, and travertine. In these cases, a relatively weak acid will begin to dissolve the surface of the stone, leaving a dull spot. Because we’re well-trained to understand etching in marble, it seems confounding that granite can suffer the same problem. Aren’t granites supposed to be acid-resistant? Well, “the dose makes the poison,” as the saying goes. A strong acid will eat almost anything, including granite. This is doubly true when the acid is in contact with the stone for a long time, as is the case with a bottle of cleaner left on the countertop.
The chemistry of the ways various minerals dissolve in acids is complex, but the bottom line is that strong cleaners can damage natural stones. Quite literally, the acid will remove some of the ions in the minerals. Other types of chemical reactions can occur too, and in some experiments, the acid-induced breakdown of feldspars formed a film of silica on the surface of the stone. Out in the natural world, naturally occurring acids break feldspars down into clay, which is a normal weathering process.
Different minerals will react with acid in different ways and at different rates. Feldspar minerals undergo chemical changes in hydrochloric acid, but quartz is generally less affected. But even different types of feldspar respond differently to acid attacks. This means that acid damage on a stone is usually not uniform. It’s likely to be speckled, with some minerals more affected than others. Minerals will show the most damage around their edges rather than in the middle. Smaller grains will be more affected than larger ones. The acid will migrate along pores and cracks, which can concentrate the damage in certain areas.
One aspect of this problem remains a mystery. Even though the minerals have undergone a change in their chemistry and color, the polished shine of the stone is often unchanged. I wish I had a scientific explanation for this, but I don’t. We’ll have to crack that nut another time.
These images show the telltale patterns of acid damage.


Now What?
Acid damage is not a stain. No amount of cleaners, poultices, or Magic Erasers will resolve the problem. The minerals have been permanently changed, and there’s no reversing it. That can be discouraging news, but it’s better to know the cold, hard truth up front.
The only way to remove acid damage is to grind it out. But depending on the depth of the damage, it may be hard to restore the stone to an even, level surface. Some people opt to “Sharpie” their way out of the problem by coloring or dying the stone to something close-ish to the original color.
Less-Damaging Products Are Available
The best way to ensure that a bottle of high-acid toilet bowl cleaner doesn’t damage your flooring or countertop is not to have it in the house in the first place. Otherwise, it’s all too easy for a guest or family member to set the bottle on the stone, causing damage, heartbreak, and potential family strife. Grist did a review of natural toilet bowl cleaners, and found some good options; their favorites were Clorox Green Works and Mrs. Meyer’s. But as always we recommend testing a cleaners on a spare piece of stone to make sure that it won’t do any damage if there is some spillage.
As always, the best medicine is prevention. Read labels carefully, buy less-damaging products, and invest in a good scrub brush so you can tackle stains physically rather than chemically. Your mother would be proud.